This blog is the script for a final video project for my Educational Technology class as a doctoral candidate at the University of Northern Colorado. The final video can be viewed at: https://youtu.be/jimJqjsetNM.
Introduction
3D modeling and printing are taking the Do-it-Yourself world by storm as makerspaces spring up in many schools. Considered to be an innovative way of learning next-generation skills, 3D modeling and printing are fun hobbies, but are they effective educational tools? Is 3D technology worth the cost and the time it takes to learn? Will a 3D printer merely sit in the corner and collect dust, or will it be frequently and effectively used to teach class concepts? Is 3D printing just another new toy or is it a pedagogically sound method for deep learning?
My name is David Black and I have taught media design and science classes for 30 years at the secondary level. I have developed multi-disciplinary projects that combine science with 3D modeling, but I lacked a theoretical framework. This video explores the history and innovation of 3D modeling and printing within a theoretical framework of constructivism and a project-based learning pedagogy to effectively teach science concepts. We will look at the diffusion of this new technology, how it works as a medium to convey learning, the basic steps and history of producing 3D models and prints, and provide examples of 3D technology use in science classrooms.

A Theoretical Framework
When students create their own science educational content, or learner-generated digital media (LGDM), they achieve a deeper understanding of the science. Researchers have found that students not only learn science content well through media creation, they also develop marketable media design and 21st century skills of collaboration, communication, critical thinking, and creativity (Hoban, Nielsen, & Shepherd, 2013; Orus, et al., 2016; Reyna, 2021).
Reyna and Meier (2018) conducted a literature review of studies that use learner-generated digital media to teach science concepts. They concluded that previous studies were limited because they lacked theoretical frameworks or sound pedagogy. These researchers assumed that the participating students already knew how to use media design technology tools since they were so-called “digital natives.” According to Reyna and Meier, just because students grow up using computers and digital devices doesn’t mean they have ever developed media creation skills such as video editing or 3D modeling. In a follow up study, Reyna scaffolded media design skills training through smaller partial projects embedded in a theoretical framework of constructivism and a project-based learning (Reyna & Meier, 2018; Reyna, 2021). As an example, a teacher might have a student team create a short Public Service Announcement (PSA) as a practice project to gain skills in using cameras, lighting, and microphones and to learn the entire video creation process or workflow before tackling the final project.

Constructivist theory proposes that learning is a socially mediated cognitive process whereby learners experience a subject and construct their own meanings for it. They create mind maps or schema that tie previous learning, emotions, and social reinforcement together with their new knowledge. Schema develop through the processes of assimilation, where the new knowledge is placed into existing categories, and accommodation, where the schema are revised to acknowledge divergent information. Constructivism acknowledges that the learner is at the center of the process.
3D modeling is inherently a constructivist activity, as creating the models literally requires constructing one polygon or primitive at a time. That the model exists only in virtual space does not mean it is any less a constructed medium. By 3D printing the virtual model, the printer builds an actual model one layer at a time. If properly planned and conceived, students can also construct science knowledge through 3D modeling and animation. Instead of consuming scientific content, students become producers of content. They become the experts and the teachers, and learning occurs as a natural byproduct of the process.


Constructivist theory can be traced all the way back to Socrates, who said, “Education is the kindling of a flame, not the filling of a vessel.” John Dewey proposed that students learn by doing, that is, in an active, creative process where they construct their own meanings through discovery with the teacher as a guide on the side, not the sage on the stage (Brau, 2020). It is the opposite of objectivism, where teachers are the center of the process and must somehow pour their knowledge into their students’ brains. Lev Vygotsky added that learning is socially mediated through interpersonal interactions, language, and culture and that students learn within their zone of proximal development; as students learn more, what they can do with help (the ZPD) expands (Brau, 2020). Jean Piaget developed cognitive constructivism where children develop naturally through various stages from concrete to abstract, with each stage of cognitive growth affecting the construction of learning (Brau, 2020). Seymour Papert developed his constructionist theory where students construct learning through making. He saw computers as a tool for learning and invention where students learn through doing and experimentation, including the use of computer programming and media design. Today’s makerspaces are based on his theories.

Project-Based Learning Pedagogy
Project-based learning is a natural fit as a pedagogy for media design creation. It usually occurs in teams and the conclusion is a public product. According to PBLworks, formerly the Buck Institute for Education, gold-standard PBL includes seven characteristics (https://www.pblworks.org/):
(1) A challenging, meaningful question or problem to address; (2) Student inquiry using authentic data or sources where they discover the learning for themselves as a natural outgrowth of the initial question; (3) Student voice and choice in the type of project chosen and how it will be accomplished; (4) Collaboration and communication as students actively participate and work through issues creatively; (5) Frequent opportunities for critique and formative feedback, with revision; (6) A public presentation of the final product; and (7) Student reflection on what they have accomplished and learned.
Each project ends with a presentation before a public audience, usually at some type of back-to-school night with feedback and suggestions from the audience. Knowing that their work will have a public audience motivates students to deliver a high quality product and helps to actively engage them in the process of learning. As John Spencer, a well-known PBL guru, explains:
Students who engage in authentic project-based learning have increased agency and ownership. They’re often more excited and engaged in their learning. When this happens, they retain the information for a longer amount of time while also learning vital technology skills like digital citizenship and media literacy. However, they also learn vital soft skills, such as collaboration, communication, curation, and problem-solving. As they work through iterations and revise their work, they develop a growth mindset. Often, they learn how to seek out constructive feedback. This connection to the community can help them develop empathy (Spencer, 2021).

The Message and the Medium
In the early 1960s, Marshall McLuhan created the concept of the global village, which predicting the interconnectivity of the World Wide Web, and famously stated that “the medium is the message.” Such technologies as print and movable type have a profound effect on the human psyche and cultural understandings. Humans re-invent themselves and how they communicate with the invention of each new medium (McLuhan, 1962). Richard Clark argued conversely in the 1990s that the medium of a message was unimportant to the learning process and that cost-effectiveness was the only consideration in choosing one medium over another in instructional design (Clark, 1994). If more than one form of media can be used for a particular learning task, then they are replaceable with each other and the medium does not influence the message. Robert Kozma (1994) takes a middle ground, similar to Kalantzis and Cope (2012), where the medium conveys or mediates the message, influencing the message because of the medium’s unique affordances (advantages, disadvantages, limitations, conventions, etc.). Learning from a video with its linear format is qualitatively different than learning from interactive media such a website or a CD-ROM-based multimedia title. Learning from printed text alone is different than learning from text with images, or moving text and images in the form of a video.

3D modeling and animation has its own affordances that allow it to present a unique learning experience unlike other media. It can visualize large datasets, allowing patterns and inter-relations between data to be understood that purely numeric data tables cannot. A chart of the stock market can lay out several indices together for comparison over time, representing over 80,000 different data points in one infographic, which would be impossible to interpret as raw data but is easily accessible visually (Krum, 2013). If the data represents values in a two-dimensional grid, then a 3D graph is the best way to visualize and understand the data, as in the example of the voltage data I will talk about later. The medium chosen to convey learning is therefore an essential part of the learning experience. This agrees most closely with Kozma’s middle ground stand on the Clark-McLuhan continuum (Kozma, 1994).
With this in mind, let us now turn to an examination of the history of 3D modeling and printing as an innovative technology and how it can be used in science classrooms.

History of 3D Modeling
The first experiments with 3D computer modeling began in the 1970s using mainframe computers, the only ones that could handle the millions of calculations necessary. A group at the University of Utah’s computer lab led by Ed Catmull developed a process to build smoothly polygonal models with accurate reflections called raytracing. As microprocessors improved and computer speeds and power increased, the first entertainment applications appeared. By the early 1980s movie studios were experimenting with computer graphic inserts for special effects. Disney’s Tron brought complete scenes designed and rendered in 3D, followed by even more photorealistic effects in The Last Starfighter with complete Codon Armadas including spaceships, planets, and asteroids.

Meanwhile, some of the artists that started with Tron founded their own studios, including Robert Abel and Associates, who created a number of iconic and Clio Award-winning TV commercials including the famous 1984 Super Bowl sexy robot commercial that introduced innovative motion capture technology (Art of Computer Animation, n.d.). Lucas Films and Industrial Light and Magic experimented with completely 3D animated short movies. When Steve Jobs left Apple Computers and invested in the studio, they became Pixar Animation Studios and created the groundbreaking Luxo, Jr. animation. Jobs encouraged John Lasseter to create a full-length 3D movie; the result was Toy Story in 1995 and the rest is history.
History of 3D Printing
In 1981, Hideo Kodama of Nagoya Municipal Industrial Research Institute published a description of a liquid resin-based photopolymer that becomes solid by hardening each layer with focused ultraviolet light, but he did not file a patent (Goldberg, 2018). In 1984, Charles Hull invented a similar system called a stereolithography apparatus (SLA). Refinements continued, including building up layers by sending UV light in cross-sections. In 1992, the first Selective Laser Sintering (SLS) device was invented, which uses a laser beam to sinter or weld together a powder into layers.

Although both of these techniques are still used in high-end industry, the type of 3D printing most familiar in schools is the Fused Deposition Modeling (FDM) technique where a plastic filament on a spool is fed into the printer by a motor, melted by a heated nozzle, and deposited in layers continuously to make cross-sections (3Dsourced.com, 2019). For each layer, the build plate is moved down. It is also called Fused Filament Fabrication. This type of printing was first developed in the late 1980s by F. Scott Crump who went on to found Stratisys in 1990 as the first company to build FDM printers and plastic filament.
Altogether, these technologies are referred to as additive manufacturing because the models are built up, or added, layer by layer. By comparison, a 3D milling machine or CNC router uses subtractive manufacturing because it starts with a larger blank and carves away parts.
By 2009, when the first FDM patents expired, new companies entered the market with lower cost desktop 3D printing machines such as MakerBot, FlashForge, Prusa, Ultimaker, Dremmel, and others. Various types of plastic filament became available, including Polylactic Acid (PLA), Acrylonitrile Butadiene Styrene (ABS), and even flexible nylon filaments, fused metallic filaments, and water dissolvable filaments for printing supports. Filaments come in many colors and finishes, including filaments that change color as they spool into the printer. A new development is 3D printing in full color, however these printers cost $3500 or more. To print in color, 4-process color (CMYK) dyes are added to a base color filament as it extrudes.

The Process: From Model to Print
Many 3D models are available for free on Thingiverse (https://www.thingiverse.com/) and other sites, but ultimately the fun of 3D modeling is to do it yourself. Complicated by working in three-dimensional space on a two-dimensional computer screen, in 3D modeling primitive objects such as spheres, cubes, and cylinders are given textures and composed into scenes. They can be combined to add, cut, or intersect other objects using Boolean commands. Other objects are created using a polygonal mesh like chicken wire. Meshes act as three-dimensional vectors and can be modeled from equations and deformed using envelopes. Grayscale images can be converted into terrain objects with light areas as mountains and dark areas as valleys.

To create complete scenes, the objects are aligned and composed, the camera positioned, lighting and atmospherics provided along with other procedural effects, then rendered out through ray tracing as if the scene were being photographed by bouncing a beam of light out from the camera. To animate objects, a timeline is added and the objects are given hierarchical links from parent to child, a process called forward kinematics. The pieces can be moved, rotated, and otherwise changed over time by adding keyframes on the timeline. The computer then renders out an animation frame by frame, creating all the in-between frames itself. Complex characters can be rigged with bones and joints or morph targets to deform the polygonal structure over time.
Learning 3D animation is traditionally a difficult and time-consuming process, with each step from modeling to rigging to texturing to animating done by different teams of specialists for a major CGI-based motion picture. Teaching students how to do all of these steps competently can take several weeks of class time, if not years, which few teachers or subjects can afford to do. However, new tools are simplifying the process.

TinkerCAD is a browser-based modeling tool that has a range of primitive objects and simple textures that can be combined into more complex models, which can then be exported and printed with a 3D printer. It is not set up to do complex polygonal modeling or animation, but is a good introduction to working in three-dimensional space. SculptGL is another browser-based modeling tool. It does create complex meshes, but instead of subdividing polygons one at a time, the tool works as a ball of virtual clay that can be pushed and pulled. The model can be colored with paintbrushes and the final models and textures exported as .OBJ files for use in more sophisticated animation software. I have not yet found a browser-based tool that can assemble complete scenes, add keyframes, rig bones and morph targets, and render out animations. There are full-scale downloadable programs available for free or as educational licenses, including Blender and Autodesk Maya. Many tutorials exist online for how to use these programs.
Once a 3D model is completed it is saved as an .OBJ or .STL file, then imported into a 3D printer’s slicing software which provides the G-code directions for moving the print nozzle across the build plane while extruding the melted filament. Where overhangs occur, supports must be built in (which can be done automatically or by hand). Once the model begins to print, it can take several hours to complete a moderately large print. The print must then be removed from the build plate and the raft and supports snapped off and sanded.
Subject Integration and Adoption of 3D Modeling and Printing in Schools
Reyna’s 2018 literature review concluded that most previous research in learner-generated digital media lacks theoretical frameworks or solid pedagogy. This agrees with the TPACK model of technology integration (Rodgers, 2018), where the affordances and workflow of the technology, the appropriate pedagogy for teaching, and how students learn the content knowledge through the technology or medium must be considered to successfully integrate technology into the classroom. By using a constructivist/constructionist framework and the pedagogy of project-based learning and by training students how to create the media as they develop their own science-related content, we are following the TPACK model.
For 3D modeling, we cannot assume that even digital natives know how to use the conventions of modeling in three-dimensional space on a two-dimensional surface. It is a challenging innovation to learn, and many problems can occur in the modeling and printing process. Students do not naturally know how to subdivide polygons, use Boolean commands to cut holes, create procedural UV texture maps, create the lighting and atmospherics needed for a scene, or set up a 3D object for a successful print. This is highly technical work, and requires practice and scaffolding with simpler projects before large-scale media projects can be undertaken. Time must be set aside for training and practice either in class or using a flipped classroom model. The purpose of the 3D modeling – to learn a science concept – must be carefully considered and needs to be worth the time and effort and cannot be adequately taught through any other medium.

Because of these challenges, 3D modeling and printing have been slow technologies to truly take hold in schools. Although 3D printers are popular now, most schools and teachers have little idea how to use them effectively. If we use Everett Roger’s model of technology adoption (Legris, Ingham, & Collerette, 2003), schools are usually in the late adopter or laggard phase for adopting 3D technologies. They do not want to waste the time or dollars to invest in a 3D printer just because it is the newest shiny thing. Some individual teachers may be ahead of the curve and ready to adopt, but they will need to learn how to use 3D technologies on their own; there is little to no professional development training available through school districts unless provided by teacher associations.
Using 3D Modeling and Printing in Science Classes
Because of the time and challenge required to do 3D modeling, printing, and animation there should be a compelling reason for using this medium in a science classroom; the 3D model or animation must convey a scientific concept more effectively than other forms of media. Some possible applications include modeling and animating scientific processes or principles, modeling complex authentic data where it cannot be visualized in any other way, and creating accurate models of science-related objects that can be examined.
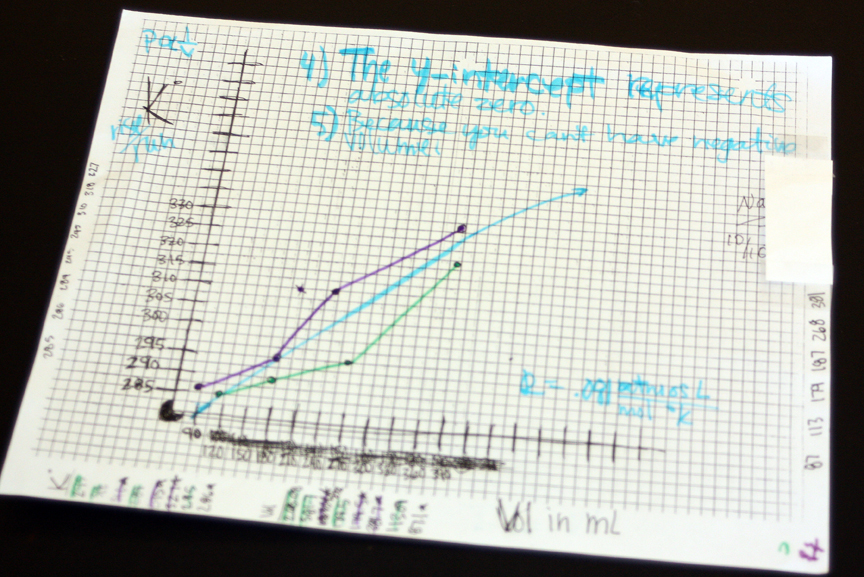
To demonstrate how data can be visualized in 3D, my chemistry students this week studied electrochemistry by comparing the voltages of different combinations of metal electrodes, recording the data in a two-dimensional grid separated by commas. We used a free program from the National Institutes of Health called ImageJ to convert the raw numbers into a grayscale image, with higher values represented as lighter shades of gray and lower numbers as darker shades. This image can be converted into a 3D object by Adobe Photoshop. Finally, an altitude sensitive texture is applied and text added and the scene rendered as an image or animation. Patterns in the data that are difficult to notice as raw numbers become readily apparent as a visual image. Students can easily see that magnesium is the most reactive metal and has the highest voltages. 3D visualization has great advantages over trying to understand a grid of numbers.
As an example of building models to illustrate concepts and objects, in 2004, my media design students began work on a video documentary for KUED, Salt Lake City’s PBS station, on the history of AM radio in Utah. They created animations of transistor radios as part of the title sequences of each segment based on photographs of actual radios. They modeled interfaces for an interactive DVD of our final video and for an accompanying CD-ROM programmed with Macromedia Director. Students in my 6th grade Creative Computing class each modeled a part of the SOFIA aircraft, or Stratospheric Observatory For Infrared Astronomy, and my high school students assembled, textured, and animated the final model. It was used to demonstrate how the telescope works for a video we made about my flight on SOFIA in 2013.

My media design students created models and animations of Mars space probes for an interactive CD-ROM on Mars Exploration which they presented at a student symposium at Arizona State University. They learned how to access and model 3D terrain data of Mars from the MOLA instrument on the Mars Global Surveyor probe to analyze possible landing sites on Mars which we then printed out using color-changing PLA filament. Other students collaborated with the NASA Lunar Science Institute to study selenographic features and create a 3D animation of the Big Impact Theory of lunar formation.
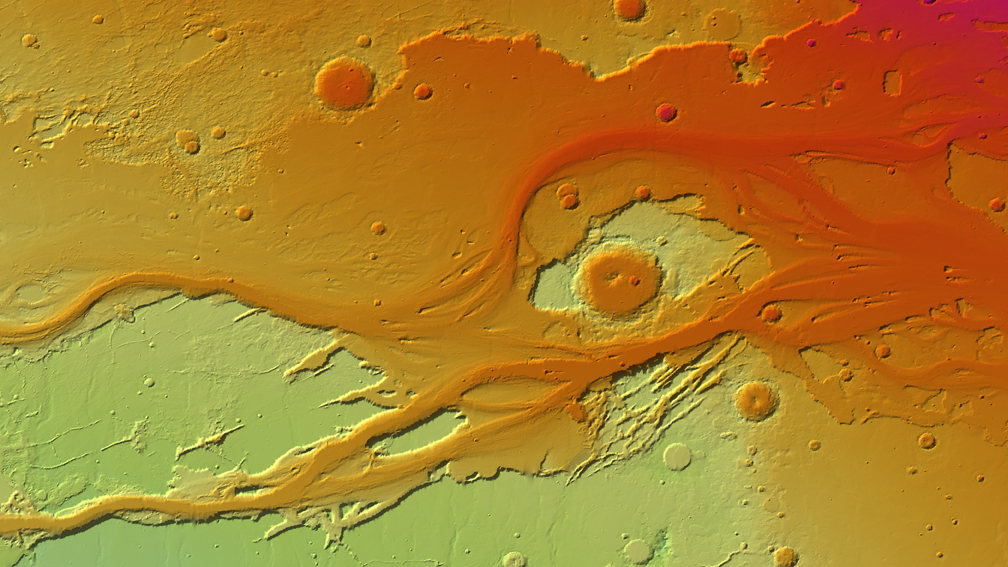
My biology students use TinkerCAD or SculptGL to model and print viruses for a unit on microbiology. To visualize the periodic properties of the elements, my chemistry students create a grid of data points and convert it to 3D models using ImageJ and Photoshop. My eighth grade physical science students created a 3D animated video with greenscreen narration showing a possible habitat that astronauts can live in on their way to Mars. To represent land forms on Earth, my students access EarthExplorer by the United States Geological Survey (https://earthexplorer.usgs.gov/), which allows grayscale heightmaps from the Shuttle Radar Topography Mission to be downloaded and converted into highly accurate 3D models.

All of these examples took time to learn and execute, so we needed to have compelling reasons for using 3D modeling that were worth the opportunity cost of time and the steep learning curve. In each case, using 3D enabled us to present authentic data more completely and helped us visualize important scientific concepts more effectively than other types of media.
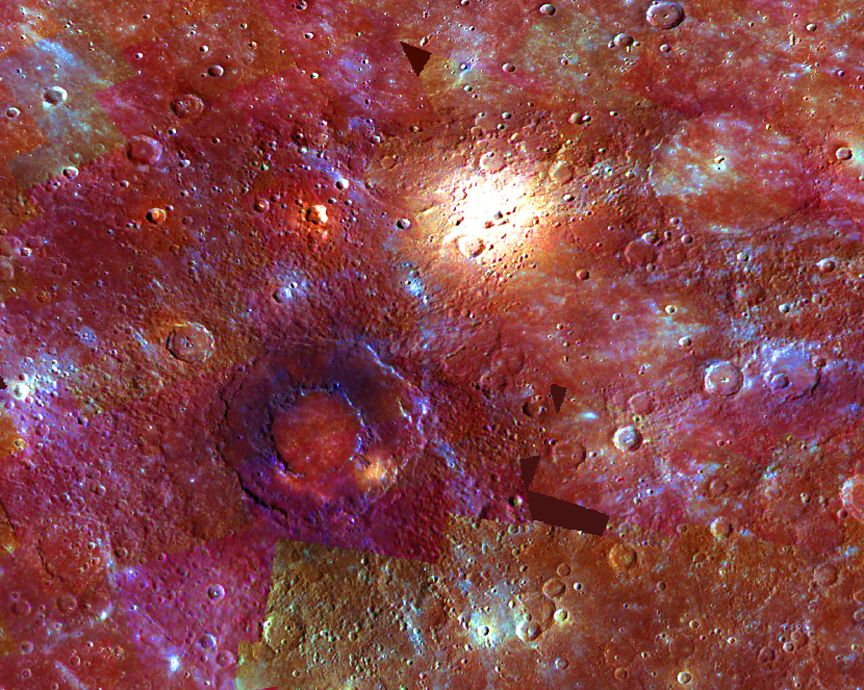
Using 3D modeling has benefits for students. Visualization of big data sets has become a growth career, as has 3D printing of everything from prosthetic limbs to cars to rocket parts. It allows for rapid prototyping and has become a necessary component for engineers. My students have used these techniques to create visuals for winning science fair projects, such as this one that determined whether surface features on Mercury were caused by impacts or volcanism. As part of the Mars Exploration Student Data Team program, my media design students downloaded Mars dust opacity data from December 2003 to January 2004 and converted it into animations showing how a dust storm arose over the Tharsis Plateau, blew across the equator, and spread globally just as the Mars Exploration Rovers were approaching. My current physics students are using altitude data from the Lunar Reconnaissance Orbiter’s LOLA instrument, turning grayscale heightmaps of the Moon into 3D models, then mapping spectroscopic data from Moon Mineralogical Mapper instrument to show where different commercially viable minerals might be located according to surface landmarks as shown here (Fa & Jin, 2007). They are creating a poster of their results for the ExMASS program to compete with students from nine other schools.

Conclusion
In this paper I have described many 3D modeling and animation projects created by my students. They have learned many positive things about STEAM careers and processes along the way, but of most importance, they learned science concepts more deeply through 3D modeling than through any other method. Although 3D modeling can be rewarding in its own right, it has additional benefits for students including teaching marketable skills and providing them with opportunities to collaborate, communicate, solve problems, and enhance their creativity. Given our limited time as teachers and the high opportunity cost, we have to be very sure that 3D modeling also enhances science learning in ways that other options can’t achieve. In my own experience, the projects are well worth their time. Other teachers will have to look at their own situations and determine whether or not it is worth investing the time to learn and use 3D modeling in their own science classrooms.
Thanks for reading this. I hope it provided some ideas into how and why to use 3D modeling and printing in your science classroom.
References
3Dsourced.com (2019). Fused deposition modeling: Everything you need to know about FDM 3D printing. Retrieved 3/18/21 from: https://www.3dsourced.com/guides/fused-deposition-modeling-fdm/.
Art of computer animation (n.d.). Retrieved from: https://youtu.be/5xwLFRdewgE.
Brau, B. (2020) Constructivism. In R. Kimmons & S. Caskurlu (Eds.), The Students’ Guide to Learning Design and Research, EdTech Books. https://edtechbooks.org/studentguide/constructivism.
Clark, R. E. (1994). Media will never influence learning. Educational Technology Research and Development, 42(2), 21-29.
Center for Educational Innovation, (n.d.). Constructivism. Center for Educational Innovation, University of Buffalo. Retrieved from: http://www.buffalo.edu/ubcei/enhance/learning/constructivism.html
Edutechwiki,(n.d.). The media debate. Retrieved 4/18/21 from: http://edutechwiki.unige.ch/en/The_media_debate
Fa, W. & Jin, Y. (2007). Quantitative estimation of helium-3 spatial distribution in the lunar regolith layer. Icarus, 190 (2007), 15-23.
Goldberg, D. (2018). History of 3D printing: It’s older than you are (that is, if you’re under 30). Retrieved 4/18/21 from: https://redshift.autodesk.com/history-of-3d-printing/
Hoban, G., Nielsen, W., & Shepherd, A. (2013). Explaining and communicating science using student- created blended media. Teaching Science, 59(1), 33-35.
Kalantzis, M. & Cope, B. (2012). Literacies. Cambridge University Press: New York, NY
Kozma, R. B. (1994), The Influence of Media on Learning: The Debate Continues, School Library Media Research, Volume 22, Number 4, Summer 1994.
Krum, R. (2013). Infographics: Effective communication with data visualization and design. John Wiley & Sons: Hoboken, NJ.
Legris, P., Ingham, J. & Collerette, P. (2003). Why do people use information technology? A critical review of the technology acceptance model. Information & Management, 40 (2003), 191-24.
McLuhan, M. (1962). The Gutenberg galaxy: The making of typographic man. University of Toronto Press: Toronto, Canada.
Orus, et al. (2016). The effects of learner-generated videos for YouTube on learning outcomes and satisfaction. Computers & Education, 95 (2016), 254-269.
Reyna, J. (2021). Digital media assignments in undergraduate science education: An evidence-based approach. Research in Learning Technology, 29 (2021), 1-19.
Reyna, J. & Meier, P. (2018). Learner-generated digital media (LGDM) as an assessment tool in tertiary science education: A review of literature. IAFOR Journal of Education, 6(3), 93-109.
Rodgers, D. (2018). The TPACK framework explained (with classroom examples). SchoolologyExchange. Retrieved from: https://www.schoology.com/blog/tpack-framework- explained.
Spencer, J. (2021). PBL for all. Retrieved from: https://spencerauthor.com/pbl-for-all/