Once each year I like to go over the statistics for this blog in detail to see what posts have been the most popular, which search terms are finding this blog, which videos are most watched, etc. I’m not doing this just for an ego trip, but to be able to report the impact this site is having. I have had some very generous sponsors over the three years this blog has been running, especially the American Section of the Société de Chimie Industrielle (which paid for my fellowship in 2009) and the Chemical Heritage Foundation, which provided such a wealth of resources in its collections on the history of chemistry. It was during the time of my fellowship that this blog really began to find an audience, and it has been growing ever since.
So here is where this blog stands: As of today, there have been a total of 67,620 visits to this site. As seen by the histogram, the number of visits has shown a definite annual pattern consistent with the school year – visits are lower in the summer when school is not in session, rise in August and September, stay high in October and November, dip a bit in December due to Winter Vacation, then rise again in January and February and peak in March, then gradually decrease as the school year winds down in April and May. This same pattern has repeated for the last three school years, but has grown each year. Last year, in the 2010-2011 school year, my best months were slightly above 3000 visits. Now they are topping out above 4000 and I hope they will hit 5000 by March.
Granted, compared to some popular blogs with thousands of hits per day, 5000 per month doesn’t sound like much. However, I am pleased – this is a rather esoteric blog dedicated to the history of chemistry and chemistry education. The yearly pattern shows that I am reaching my intended audience of high school students and teachers. This is also shown by the types of searches that reach my blog.
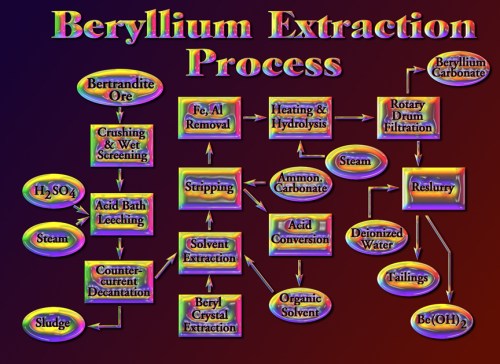
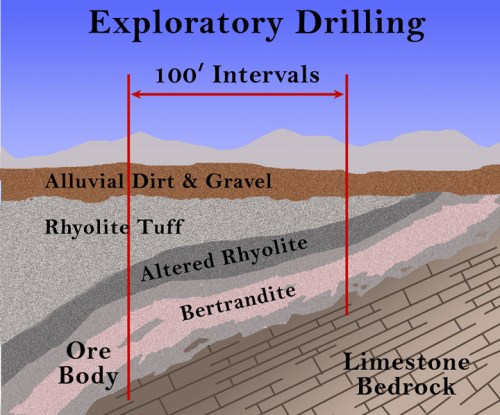
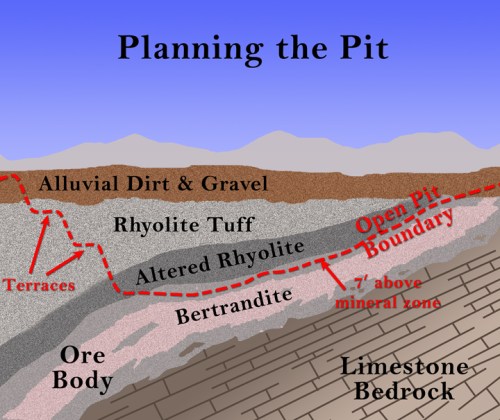
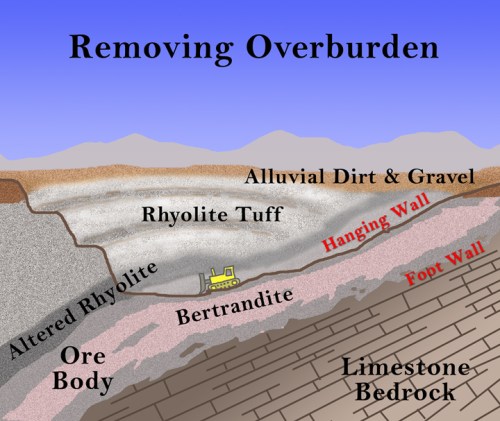
Although there are always some unrelated search terms that somehow reach my blog (the biggest ones are “Ocean City, New Jersey” and “Punxsatawney Phil” because I visited both places in 2009 and showed some pictures), by far the majority of search terms are related to chemistry and its history or to science education in general. I’ve gone through the search terms and compiled them into categories, mostly so that I can make plans for the future. Here are the top searches that reach this blog: (1) Greek Matter Theories (3473 searches) with Aristotle, Democritus, and Thales being the biggest ones; (2) the Periodic Table of elements (2288); (3) beryllium (1600); (4) Alexandre Beguyer de Chancourtois (1397) – this is a bit surprising, but apparently my animation of his telluric screw periodic system and description of his work is one of the few sites out there about him; (5) the Tintic Mining District (1041); (6) the history of the periodic table (868); (7) science education (862), especially using iPads in science classes; (8) early modern chemistry (822), including Lavoisier, Boyle, Priestley, Dalton, and Newton; (9) alchemy (732), with love potions, Khunrath, Basil Valentine, Zosimos, and Maier the highest; (10) water and wind turbines (618); (11) strange attractors (586) – this is another odd one, since I only mentioned it once, but it was in my most popular post; (12) mercury (554); (13) early technology (514), such as Roman glass, Pliny the Elder, Agricola, Neri, and others; (14) mining in general (455) – such terms as overburden, open pit mine, ball mill, and headframe; and (15) Cripple Creek, Colorado (315).
The videos that I have created for this project are posted on this blog (under the video tab) and on YouTube. The History of the Periodic Table, featuring Dr. Eric Scerri of UCLA, is my biggest hit so far. All parts of this video have been watched a total of 11,474 times as of 1/7/2012. There are even a few derivative works on YouTube that take parts of my video – a section on Henry Moseley, for example – and combine it with parts of other videos with Bill Nye, etc. I’ve had quite a few comments on how useful this video has been for chemistry teachers out there, and I am very pleased with the results so far. There is also a version with Portuguese subtitles done by a professor in Brazil; I’m not sure how many times that has been seen. My separate video that showed only some animations of the periodic table has been watched 416 times.
The second most popular videos have been the two parts on beryllium – its properties and uses, and how it is mined and refined. It has been watched a total of 3219 times, with the separate video on the geology of beryllium watched itself an additional 153 times. The Discovery of Synthetic Diamonds has been watched 745 times and the demonstration of Glass Blowing 754 times. These have been the most popular videos related to this project.
In conclusion, the most important question is: Have I succeeded in my attempt to bring the history of chemistry and chemistry education to the general public, and specifically to teachers and students? All indications, based on these statistics, are that I am succeeding and that that success is continuing to grow.
The last several posts have been about astronomy and space science education, and although some search terms have reached these posts, not many have. For various reasons, not the least of which is that I want to keep this blog focused on my original intent, I am starting a new blog which should be up and running by Wednesday night on space science education and resources for teachers to use now that we are in the golden age of astronomy. I will be doing quite a bit of education outreach on these topics over the next few years, if all goes well, and they deserve to have their own blog. I will include links here once that is ready to visit. I will post to this new blog once per week on Wednesdays.
The statistics also point out which topics have been most popular, and give me direction on what to post about in the future. In my next post, I will give you a schedule of what I intend to discuss over the next year and a half and when I will have the related videos completed. I will try to post once per week, probably on weekends. I have much more material from my fellowship at the Chemical Heritage Foundation that I haven’t shown or discussed here yet, and I look forward to digging into it all. I have also visited many sites related to mining and refining of the elements which I have only mentioned in passing. It’s time to edit all that footage and photos into videos for this site and YouTube. I expect the next few years to be busy, productive, and rewarding and to reach even more people than I already have.