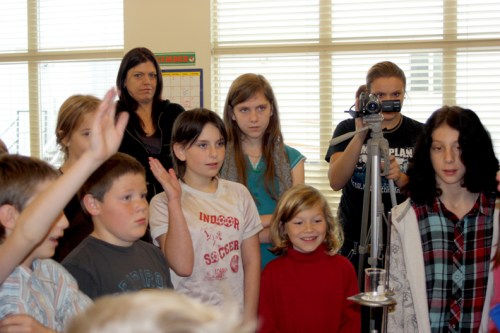Last December right before winter break, my chemistry students prepared demonstrations to present to each other and to the elementary classes here at Walden School. This was their first attempt at it, and they received evaluations from me and from their peers with suggestions on how to improve. Now we have just finished the second round of presentations, and each team has added new features and made improvements.
I had each team improve their presentations in four areas: first, their presentation skills, such as speaking with good diction, showing enthusiasm, and having a smoothly scripted and rehearsed narrative. The second area was improving the visual appeal of their presentation by adding some sort of poster or handout that could be used as an activity for the audience while the team sets up. Some of the groups made posters, some made paper games such as word searches or worksheets. The third area to improve was to add a multimedia component, such as a powerpoint slide show, a video, or a game. The final area for improvement was to make their presentation more hands-on for the audience, such as having more audience participation, or some sort of kinesthetic activity, or turn the presentation into an inquiry-based lab.
The results were very good; all the presentations have improved. Their science content was already good, but is deeper and more engaging now. By adding slide shows, posters, games, activities, and participation, they have gotten their audiences much more involved and excited.
Here are some examples: One group presented on the properties and uses of silver, and their demonstration was how to untarnish silver. They not only had a good slide show, but created a kinesthetic activity where the elementary students linked arms to form first silver sulfide (tarnish) and aluminum, then reformed to create aluminum sulfide and pure silver. This demonstrated the idea of conservation of matter in chemical reactions.
The cabbage pH group turned their presentation from a demonstration into an inquiry lab by pouring samples of many types of household chemicals and food (such as grapefruit juice) into small clear plastic cups, then having the elementary students predict whether the chemicals were acids or bases, then use the cabbage juice to prove their guesses.
My favorite improvement was in the saltwater density group; they had some difficulty during their last presentation with not having practiced enough and having things not work out as planned. This time it went smoothly, and they even created a computer video game called Salt the Slug. Jess created the graphics and Josh did the programming. The purpose of the game is to use the trackpad of the computer to shake a salt shaker up and down, shaking out salt onto a slug that is crawling across the screen trying to steal food. If the player can kill the slug before it gets back to its home with the food, he or she wins. Yes, the concept sounds a a bit cruel but it taught the idea of osmosis and concentration of solutions and besides, the graphics were hilarious. The elementary students were jumping up and down for a chance to play, so the team had to ask them some review questions to decide who would get a chance to try the game out.
Josh has become an excellent game programmer and created another game, which he has been working on for a year, where the player places towers that then shoot into a maze to repel invaders. He presented this game at the Charter School Science Fair for all of central Utah, and now has qualified to go on to the regional science fair at BYU in late March. I was a judge at the fair last week, and it was amazing to see the caliber of some of the projects.
One of my favorite things about doing these presentations is that many of my high school students have younger siblings in the elementary classes; what better way for my students to show off what they’ve learned, and how they can do science, than in front of their younger brothers and sisters? Dallas, one of the students in the group that demonstrated gunpowder had his little sister in the class. They kidded each other a lot, and Dallas had to tell her, “Don’t get sarcastic with me, I taught you sarcasm!” This group also included a nice demonstration of the “toast the runt” reaction, where potassium chlorate is heated to start it decomposing and giving off oxygen, then a runt candy is rolled down the test tube as the fuel source, resulting in a stationary solid rocket motor.
We had to reform some of the groups, since a few students had switched sections at the semester, but the same presentations continued. Those teams that presented to their peers last time presented to the elementary classes this time, and vice versa. Each team has now presented twice and received feedback. Now they will present one more time at the end of the year at our Mad Science Night, where their parents and siblings are invited and we will take over four classrooms and run simultaneous sessions. It will be a lot of fun, and their presentations will be amazing.
Meanwhile, it has been quite a bit of time since my last post. I haven’t been ignoring it; rather, I’ve been so busy teaching, grading, entering competitions (such as the Explore Mars competition I mentioned previously), creating some video projects on the side for clients, presenting at the Utah Science Teachers Association conference (the Mars lessons again), and preparing for my trip to the NSTA conference next week that I simply haven’t had a chance to do many blogs. However, I have quite a backlog of student written blog posts that I will be adding over the next week, then posting each day from San Francisco, so you’ll see quite a few posts this month.