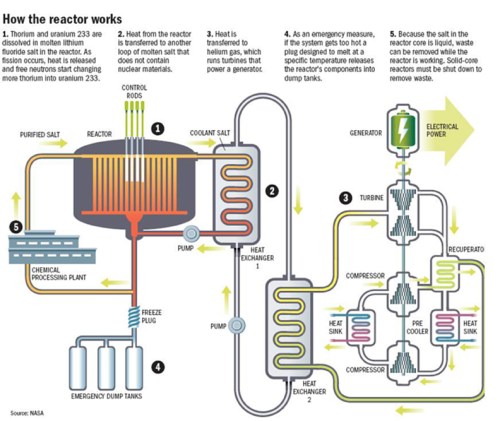
As we have studied the history of chemistry for our recent unit in Honors Chemistry, I’ve had my students do a bit of research on what is known and supposed about various alchemists. For example, a student in each of my sections was assigned to research Ramon Llull, the Majorcan alchemist. We started by finding out what is known about the real person. He was born in Palma in 1232 AD, and was a courtier, poet, and womanizer at the court of King James of Aragon, then had a religious epiphany that converted him into a fervent missionary for Catholicism. After a nine-year hermitage and writing many religious tracts, he set off on a series of missionary journeys to North Africa. He was fluent in Arabic and was unusual for his time in that he believed in converting the Muslims through reasoned argument instead of Crusades and the sword. He wrote some of the first works in Catalan, his native language, and died after being stoned in Tunis.
I also had the students research what is attributed or credited to the person in tradition and later writings, such as Ramon Llull’s alchemical works and his having created the Philosopher’s Stone.
Each student also had to find an image of the person and include it, then take their short report and convert it to simple bullet points to summarize their findings. I’ve now taken those bullet points and turned them into a Keynote/Powerpoint slide show and added their images as well as photos I took last year at the Chemical Heritage Foundation as part of my fellowship sponsored by the Société de Chimie Industrielle (American Section). This is the first time, except for a few progress report blog posts, where I have started to use all the materials I assembled. I am attaching it here, and hope you enjoy going through it and using it in your own classes.
Alchemy_History (Powerpoint)
Alchemy_History (PDF)
It was my privilege last summer to dig into the very books these alchemists wrote, and I’m still digesting what I discovered. One result has been my own creation of the White and Red Elixirs and the formation of the Stone itself; in fact, I demonstrated my alchemical prowess for my students by converting copper into silver and then into gold. Several of my students had achieved the inner transmutation sufficiently to successfully direct the Stone’s powers as well, as shown in the photo. (Of course, we really aren’t making gold. This is the old “Alchemists Dream” activity where copper pennies are coated with sodium zincate [using a combination of 6.0 M sodium hydroxide and zinc powder], then heated gently in a Bunsen burner flame to alloy the zinc with the copper to form brass, which looks like gold).
These student-created projects are part of my overall philosophy of science education and the main rational of this Elements Unearthed project: that students learn best when they are actively involved in sharing their knowledge with others. With modern tools for publishing on the Internet through blogs and PDF files, Powerpoints and videos, students now have an audience for their work that is much greater than simply their peers and teachers in class. Tomorrow is the unit test; we’ll see if my theory holds water then!