Dyed merino wool yarn using natural dyes. Top left: Rabbitbrush. Top right: Cochineal treated with ammonia. Bottom right: Indigo. Bottom center: Cochineal treated with citric acid. Bottom left: Madder root.
As a follow up to our inquiry lab to develop the best formulas for dyeing cloth with natural dyestuffs, I ordered some Kona 100% merino wool yarn and several yards of untreated cotton fabric from Dharma Trading Company along with indigo, cochineal, sandalwood, and madder root dye powders, and some mordants and other chemicals needed for these dyes.
As we finished up before Winter Break, I started testing these dyes and experimenting with variables to get an initial feel for how well the yarn and cotton work. My first test was rabbitbrush, as I had collected boxes of flowers before the color completely faded in October. I simmered a skein of yarn in aluminum sulfate (alum) powder as a mordant for an hour while boiling the rabbitbrush blossoms, then transferred the hot yarn into the dye bath. It accepted the color nicely.
Next came madder root. I used the same mordant bath and prepared a dye bath by soaking the madder root bits directly in hot water and letting it simmer while the yarn was in the mordant bath, then filtered the madder solution through a sieve before dyeing the yarn. The color did transfer, but was lighter than I had expected but a very nice light salmon orange. I used the same solution for about two feet of the cotton, but it turned out even lighter. Increasing the concentration of the dye bath didn’t seem to help.

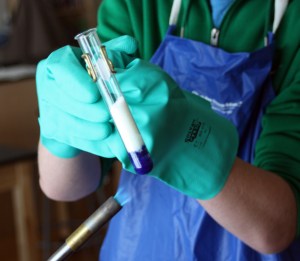
Dyeing With Cochineal: The dye bath is bottom left. I crushed the cochineal shells in a mortar and pestle, then placed them in the sieve (top center) and boiled in the hot water. The yarn is simmered in the mordant (alum powder – to the right), then simmered in the dye bath, then rinsed out (in the sink in center).
With some confidence that the wool yarn was working well, I crushed some cochineal shells in a mortar and pestle and placed them in a sieve and the sieve into boiling water to make the dye bath. This was to prevent the shells from sticking to the yarn, which would have been hard to get off. I wanted to make a multi-colored skein, so I dyed part of the skein in plain cochineal, then added citric acid to the dye bath which made it turn bright red – the citric acid worked much better than the vinegar or tartaric acids had. It made a skein that varied from deep red to burgundy color. The color stuck to the yarn extremely well.

Dyeing cotton cloth in cochineal treated with citric acid (orange) and ammonia (red to purple). Unfortunately, these colors were not colorfast. Upon rinsing, they changed back to neutral pink.
I then took the same cochineal bath (it was quite strong) and added ammonia to turn it from red to purple, again making a variegated skein. I divided the bath in two and had part of the skein simmer in the purple, part in a pot with more citric acid added back. I think I diluted it too much. Part of the skein between the two pots didn’t get much dye and remained a lavender color. The final skein varied nicely from lavender to burgundy to magenta to purple. The cotton swatch I tried was left in the citric acid side (which was now orange) over a weekend and it looked nicely orange when I took it out, but the differences in color washed out when I rinsed them – the pH neutralized. I need to figure out a way to set the color in cotton, maybe by not rinsing it before placing it in a drier. The wool yarn retained the varied colors nicely upon rinsing and washing in the laundry.

Skeins of dyed yarn before untangling. Some skeins were dyed a solid color, others were variegated.
Then I tried the tricky one – indigo. I had purchased the sodium hydrosulfite, used to reduce the blue indigo to the leuco state where it dissolves and penetrates the cloth. I followed the suggested steps from my research, but ran out of time to finish the process as a fire system sprinkler pipe burst outside the school and we had to evacuate while the fire department came to fix it. I turned off the hot plate quickly and grabbed my stuff, because it was the end of the day before Winter Break. I didn’t want to wait for the all clear, so I just went home. It took me a few days to get back to school, what with preparing for Christmas and shopping, cleaning, and cooking sugar cookies with my sons, etc. The yarn and cotton had been soaking for days. By the time I rinsed everything out, the cloth and yarn were a deep blue. I think I used to much indigo powder – this stuff is strong. The cloth washed out to a light blue and after washing the yarn, it faded as well but had a nice variegated color scheme.
After Winter Break and during the start of my second semester STEAM class, we tried out one more skein dyed with walnut shells and marigold flowers. I had some marigold blossoms I picked off my flower patch right after the first deep freeze in December and had dried them out. It died the wool a golden yellow, but I tried variegating the skein using walnut shells and hulls, but the brown color washed out to an ugly tan in both the cotton and the wool yarn. A student brought in black walnuts, but the result was the same after several attempts. I tried concentrated madder dye on part of the skein, but it didn’t work well, either. I think the marigold prevents other dyes from overdyeing. Perhaps other mordants would work for the walnut. It never got as dark as I expected. So the marigold skein is my least favorite – kind of a dirty yellow. More experimentation is needed here.

Experimenting with marigold dye (middle), madder root (right), and walnut shells (left). If the colors had remained this intense, it would have been OK. But the walnut shell and madder rinsed out and were much lighter upon washing.
I met Katie Wirthin, an education specialist from the Natural History Museum of Utah, when I was presenting my STEAM session at the NSTA STEM Forum in Denver last summer, and she asked if I was interested in teaching a workshop at the museum this year. We had communicated back and forth all fall, and once I finally had my Teachers for Global Classrooms online class done (more on this in a later series of posts), I was able to teach a workshop at NHMU. The week I was scheduled to teach it to about 23 teachers, they had a power outage and had to postpone the class for a week. The next week only eight people came, but it turned out well. Katie had gotten all the materials and as usual I tried to do too much in the two hours. We did marbled paper, iron gall ink (except I forgot to bring the tea bags – they were able to scrounge some green tea in their cafeteria which actually worked far better than the regular brown tea – you could really see the black pigment form). The final activity was dyeing cloth – we used terry cloth swatches, and it worked well but we ran out of time. She still has much of the supplies left, as it was designed for more people. We will probably run the workshop again on a Saturday for three hours.

A student dyeing a swatch with sandalwood dye using a tin (II) chloride mordant. Notice the dark orange color.
Now that I have six skeins of yarn dyed, my wife has untangled it all and rolled it into balls so she can crochet a sweater from it. I’m not sure if I want the marigold color or not, but experimentation is part of this process. It might be an epically ugly sweater, but I don’t care. I will wear it proudly.

Some green dye extracted from spinach leaves.
My STEAM students are beginning the lab again, and one student is using sandalwood for the first time. She used tin (II) chloride as a mordant, and the color turned a deep orangish brown, so as soon as I get more skeins of merino wool yarn, I will dye one with sandalwood. Another one is using spinach leaves for a green dye, and we’ll see how that goes. We need to order elderberry plants or leaves for another green color (it might take a while to grow the trees), and logwood for purple to black. There is still so much to experiment on before I post the final recipes. We still have to figure out how to improve the walnut shell dye. But we’ve learned a great deal so far, and I’ll report on my second semester class in a few weeks as we continue to experiment. This is what inquiry is all about.

The skeins untangled and rolled into balls for crochet. My wife will make me a sweater from these. The cotton swatches will be turned into a patchwork quilt of our school logo.

Skeins of dyed merino wool yarn. Clockwise from top left: Cochineal treated with citric acid (red), rabbitbrush (yellow), indigo (blue), cochineal treated with ammonia (purples), and madder root (orange).