I am continuing this series of posts on the sources, mining, and refining of beryllium ore. I am in the middle of editing the interview my students did in Dec., 2007 of Phil Sabey at the Brush Resources’ Delta Concentration Mill and will have the final videos done by next week. Today I’ve been creating a series of Flash animations showing the geologic history of the Spor Mountain area where the bertrandite deposits are located. Today’s post will be on the refining process used at the Delta Mill to concentrate the bertrandite and beryl ore into beryllium hydroxide.
Refining Beryllium Ore
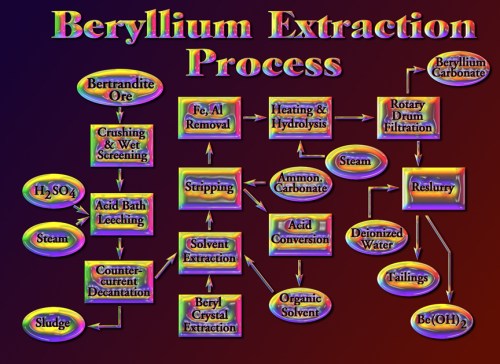
With only 0.65 % beryllium oxide (or 4.5 lbs. per ton of beryllium) in the final ore, a process had to be engineered to economically concentrate the beryllium for final processing. The properties that make beryllium useful also make it difficult to extract from its ores. Robert Maddox, Howard Gimperline, Jack Valliquet, Richard Shank, and other chemical engineers at Brush Wellman’s plant in Elmore, Ohio in the early 1960’s devised a unique solvent extraction process. With refinements, the process was seen to be economical and the go-ahead was given to build a concentration plant as close to the mine and to railroad transportation and a good water source as possible. In Dec., 1967 a groundbreaking ceremony was held at the mine and in April, 1968 a ceremony was also held at the mill site north of Delta, Utah. By the end of 1969, the plant was producing its first beryllium hydroxide concentrate.
The solvent extraction process removes the beryllium by first crushing and wet grinding the ore in a ball mill, then leaching it with sulfuric acid and steam in rotating tanks at 95 ° C to dissolve the beryllium. Thickening agents are added which help to settle the sludge in a series of flotation tanks while leaving the beryllium sulfate in solution. The sludge is stirred by counter current decantation and pumped from tank to tank as the dissolved beryllium sulfate is washed over the side to continue the process. The remaining sludge is finally discarded to a tailings pile.
The beryllium is then separated from the sulfate using an organic compound, then stripped from the organic by ammonium carbonate. Impurities of iron and aluminum are removed through steam hydrolysis, which leaves the beryllium in the form of beryllium hydroxide, which is vacuum drum filtered. Since beryllium dust is toxic, this entire process must be done in a sealed system, including the final packaging of the beryllium hydroxide into blue drums for shipment.
There are a lot of impurities in the bertrandite ore; some that gave problems early on were the high sodium content, the high uranium content, and the zirconium. The leftover filtrate still has appreciable quantities of uranium, so it is pumped to evaporation ponds, then shipped elsewhere for final uranium processing.
Once it was proven that this process could compete economically with the beryl extraction process already being used, the go-ahead was given to build the Utah processing plant. A site was selected near the Union Pacific railroad tracks and the Sevier River north of Delta and south of Lynndyl in west central Utah. The plant was completed in 1969 and began processing ore that had already been mined and stockpiled. Brush Wellman was awarded the prestigious J. C. Vaalor Award for Chemical Engineering in 1970 for the implementation of this process. In 1978, an addition was built on the plant to allow the processing of beryl ore, making the Delta plant the only facility in the United States that processes either form of beryllium ore. When beryllium was identified by the U. S. government as a strategic metal for its critical uses in the aerospace industry, beryl ore was purchased from mines in Brazil and stockpiled. Brush Resources has now purchased this strategic stockpile and is extracting the beryllium from it.
To recover beryllium from beryl crystals, the crystals must first be destroyed, since the beryllium is tightly bound in the beryl crystal lattice. The beryl is melted at 1700 ° C in a furnace, then quenched rapidly in water to break the crystal lattice and turn the beryllium particles into a frit, with the non-beryllium materials removed as slag. The frit is heat-treated at 1000 ° C in a rotary kiln, ground up in a ball mill, and leached with steam and sulfuric acid at 325 ° C in a rotating drum to dissolve the beryllium. This solution is added to the bertrandite solution in the flotation tanks to continue the process. In 1980, additional flotation tanks were added to accommodate the beryl solution.
All of these processes require careful control and monitoring to improve yields and ensure safety. Using a Continuous Improvement Process, the Delta plant has added computer automation controls and improved laboratory analysis. New flocculent agents and organic solvents have improved the extraction yields, and the plant now processes ore at a 99% efficiency level. Around 400 tons of bertrandite and about 10 tons of beryl ore can be processed per day at the Delta plant.
Special thanks go to Phil Sabey for the tour of the Brush Resources plant and for providing the brochures, Powerpoint presentations, and photos upon which this post is based.